Boiling Temperature at Altitude Calculator
Estimate campsite boiling point from elevation, pressure, and solution strength with a pressure-aware cooking model.
🏔️Camp Presets
⚙Boiling Inputs
📊Boiling Constants Grid
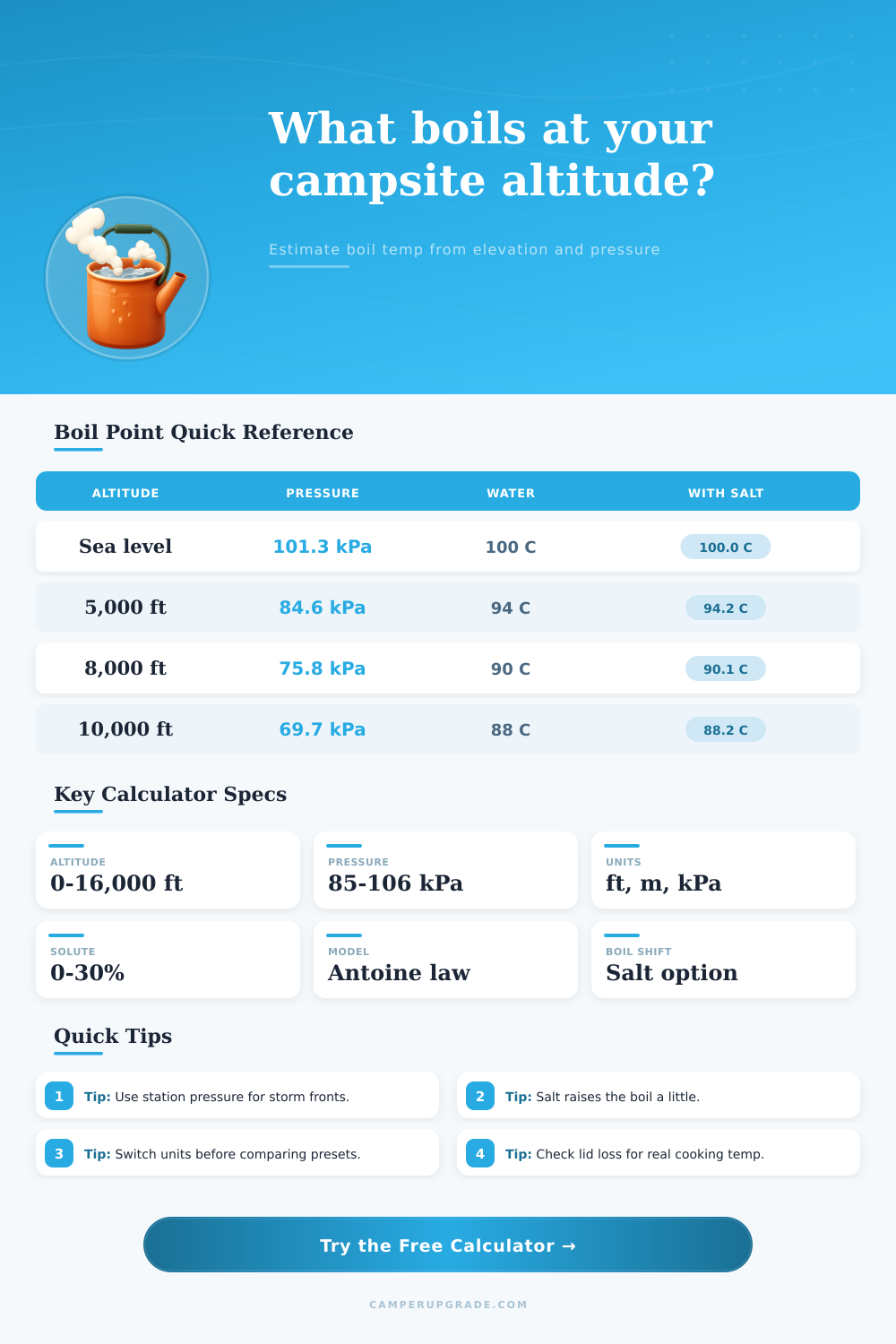
📘Reference Tables
| Altitude | Pressure | Pure Water | Notes |
|---|---|---|---|
| 0 ft / 0 m | 101.3 kPa | 100.0 C | Sea-level baseline |
| 3,000 ft / 914 m | 90.0 kPa | 96.6 C | Light boil drop |
| 6,000 ft / 1,829 m | 81.0 kPa | 93.3 C | Common mountain camp |
| 10,000 ft / 3,048 m | 69.7 kPa | 88.7 C | Longer heat-up time |
| Pressure | Boil Temp | Temp Drop | Use Case |
|---|---|---|---|
| 101.3 kPa | 100.0 C | 0.0 C | Standard boil |
| 95.0 kPa | 98.0 C | -2.0 C | Low ridge camp |
| 85.0 kPa | 94.3 C | -5.7 C | Moderate altitude |
| 75.0 kPa | 90.0 C | -10.0 C | High basin camp |
| Solute | Molar Mass | i Factor | Comment |
|---|---|---|---|
| Pure water | 0 | 0.0 | No shift |
| NaCl | 58.44 | 1.9 | Table salt |
| KCl | 74.55 | 1.8 | Potassium salt |
| Sucrose | 342.30 | 1.0 | Sugar syrup |
| Camp Style | Altitude | Boil Temp | Field Note |
|---|---|---|---|
| Lake shore cook | 500 ft | 99.1 C | Near sea level |
| Denver breakfast | 5,280 ft | 94.7 C | Watch boil time |
| Alpine pass | 9,000 ft | 89.8 C | Use a lid |
| Snowline camp | 12,000 ft | 85.1 C | Expect slower boil |
Boiling occurs when the pressure of the vapor that emerges from the liquids equals the air pressure. At sea level the temperature at which water reaches this vapor pressure is 100 degrees Celsius. This temperature is often used as a standard benchmark in cooking.
As a person climbs to higher altitudes the air becomes thin or less dense. This thin air contains less air pressures. Consequently, the boiling temperature of water decrease as air pressure decreases.
What Changes the Boiling Point of Water
Because boiling temperature determines the cooking power of the water, lower boiling temperatures mean less cooking power for the water. At elevations of 10,000 feet, for instance, water reaches its boiling point of approximately 88 degrees Celsius. Weather also plays a role in the boiling point of water.
Changes in air pressure due to weather influence the boiling point of water. For instance, weather system that feature lowering air pressures, such as storm fronts, will lead to a drop in the boiling point of water. High air pressures feature in weather systems that will raise the boiling point of water.
Meteorologists utilize handheld barometers or mobile applications for measure the air pressure at a weather station. Knowing the air pressure at a station allow individuals to adjust for changes in weather pressure. Thus, individuals should account for both there altitude above sea level and the weather pressure at there location.
The substances that are dissolved in water can change the boiling point of water. However, the effect of dissolved substances upon the boiling point of water is less than the effect of altitude upon the boiling point. For instance, boiling points of water increase if substance like salt are added to the water due to the phenomenon of colligative properties.
Salt raises the boiling temperature of water because the salt particles makes it harder for the water to reach vapor pressure. If you add 2% salt by weight to the water, the boiling point will increase by half a degree. Sugar also increases the boiling temperature of water.
However, sugar does not increase the boiling point of water as effectively than salt because it does not ionize in water. Wind and the use of lids can also have an effect on the heat in a pot of boiling water. If wind is present, it can remove heat from the pot.
Heat can drop by 5 degrees or more due to the effect of wind. Using a lid trap heat inside the pot. By using a lid, heat will not escape the pot.
If a lid isnt used, the water will lose heat more quick. Depending on the food being prepared, each food has its specific temperature needed to cook. If the boiling temperature of the water is low, food that requires a certain amount of heat will need to be cooked longer.
For instance, pasta must reach internal temperatures above 85 degrees Celsius for the pasta to properly hydrate. If the water has a low boiling temperature, the pasta will have to be stirred more or boil longer. The presence of bubbles does not mean that the food has reached the required temperature.
The appearance of bubbles only means that the food is vaporizing but does not mean that it has reached the required internal temperature. Proteins will denature more slowly if the food is below 95 degrees Celsius. Therefore, a thermometer will have to be used to ensure that the food has reached the proper temperature.
By understanding how air pressure affects the boiling point of water, how salt and sugar affect the boiling point of water, and by understanding the effect of using lids on pots of boiling water, individuals can successfuly cook foods at high altitudes.